Folks,
Richard asks:
Dear Dr. Ron,
Recently we had a solderability problem with tin-finished component leads and SAC305 solder paste. One of our engineers claimed that the problem was that the tin finish melts at too high a temperature (Tm= 232°C) for the SAC305 solder paste (Tm = 219°C) to melt it.
My understanding is that certainly above 232°C both will melt and form a good solder joint, but even if the temperature was less than 232°C, say 225°C, the tin would melt. Can you explain this phenomenon?
Richard,
Thanks for this question, which can be interpreted two ways. The first would be that, in a reflow oven at temperatures above the melting point of both metals, the one with higher melting temperature prevents the metal with a lower melting temperature from melting it. This is not true, since both metals would come near to the temperature of the air in the reflow oven and melt.
The other perspective would be that the temperature in the reflow oven is above the melting temperature of SAC305, but below that of tin. So, how can the tin melt? To consider this situation let’s say the oven is at 228°C. Will the tin on the lead or pad finish melt? The answer is yes. But, let’s try to understand the phenomenon with gold and tin first.
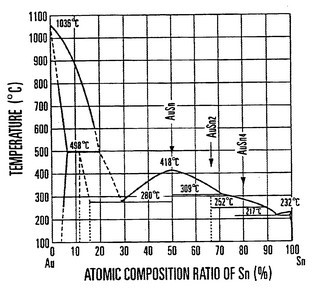
Metals that have extreme melting point differences often dissolve in each other. As you stated, tin melts at 232°C, whereas gold melts at 1064°C.
This phase diagram can be found here.
Figure 1. The gold tin phase diagram
To make a gold-tin solder, all one has to do is have a bath of tin at some moderate temperature, say 350°C. Insert the gold and the gold will melt and flow into the molten tin. This is true even though the gold melts at 1064°C. This effect can be shown experimentally. A similar phenomenon exists with gold and mercury. Mercury reacts with gold at ambient temperatures. The phenomenon can be used to extract tiny gold particles from soil and is commonly used today in artisanal gold mining. Unfortunately this use of mercury is often toxic to the miners and pollutes the environment.
Considering electronics assembly solders again, let’s assume that some liquid tin-lead solder is heated to 200°C. See Figure 2a. As seen in this figure, a ball of tin at 25°C is held above the molten tin-lead solder. The ball of tin is immersed into the molten tin-lead solder in Figure 2b. The tin-lead solder forms a meniscus around the solid tin. Even at room temperature the tin atoms are vibrating, and as a result, some of these atoms on the tin ball will end up flowing into the tin-lead solder. This action will leave a vacancy in the tin ball that may be filled by a lead atom from the tin-lead solder. In the vicinity of the newly arrived lead atom, the melting temperature of this micro spot of tin-lead alloy will be lowered as tin-lead solder has a melting temperature below that of tin. This process will continue until all of the tin will intermix with the tin lead solder and flow into it as seen in Figures 2c through 2f.

Figure 2a

Figure 2b

Figure 2c

Figure 2d

Figure 2e

Figure 2f
A video showing an experiment that demonstrates SAC solder flowing into tin-lead solder at 208°C (about 10C° below the melting point of SAC solder) is shown below. Thanks to Mario Scalzo for this video.
Cheers,
Dr. Ron